In Vivo CAR-T Development
In Vivo CAR-T Cell Therapy Services
Chimeric Antigen Receptor T cell (CAR-T) therapy has transformed the treatment landscape for cancer. Traditional ex vivo CAR-T approaches, while effective, are limited by high costs, long manufacturing timelines, and accessibility issues.
In vivo CAR-T is the next evolution—offering a transformative approach where CAR-T cells are generated directly inside the patient’s body. This eliminates the need for cell collection and laboratory modification, simplifying treatment and expanding patient access.

Key Advantages of In Vivo CAR-T
- Off-the-shelf therapy – no need for personalized cell manufacturing
- No lymphodepletion required
- No risk of graft-versus-host disease (GvHD)
- Reduced immune rejection of engineered cells
- High scalability – treat hundreds of patients per production batch
- Streamlined patient experience – simplified treatment workflow
- Compatible with multiple cell types – T cells, NK cells, macrophages
- Lower complexity and cost – bypasses ex vivo processing
Service Offerings
Non-Viral Platforms
- Lipid Nanoparticle (LNP)–mRNA/Circular RNA
- LNP–Bridge RNA
- LNP–Transposon
- Virus-Like Particles (VLPs)
Viral Platforms
- Lentiviral Vectors (LV)
- Adeno-Associated Virus (AAV)
Cell-Specific Targeting
- CD3⁺ T cells
- CD4⁺ and CD8⁺ T cell subsets
- NK cells
- Macrophages
Platform Enhancements
- Fusogen mutant screening & discovery – for enhanced gene delivery
- T cell–specific promoter optimization – to boost selective CAR expression
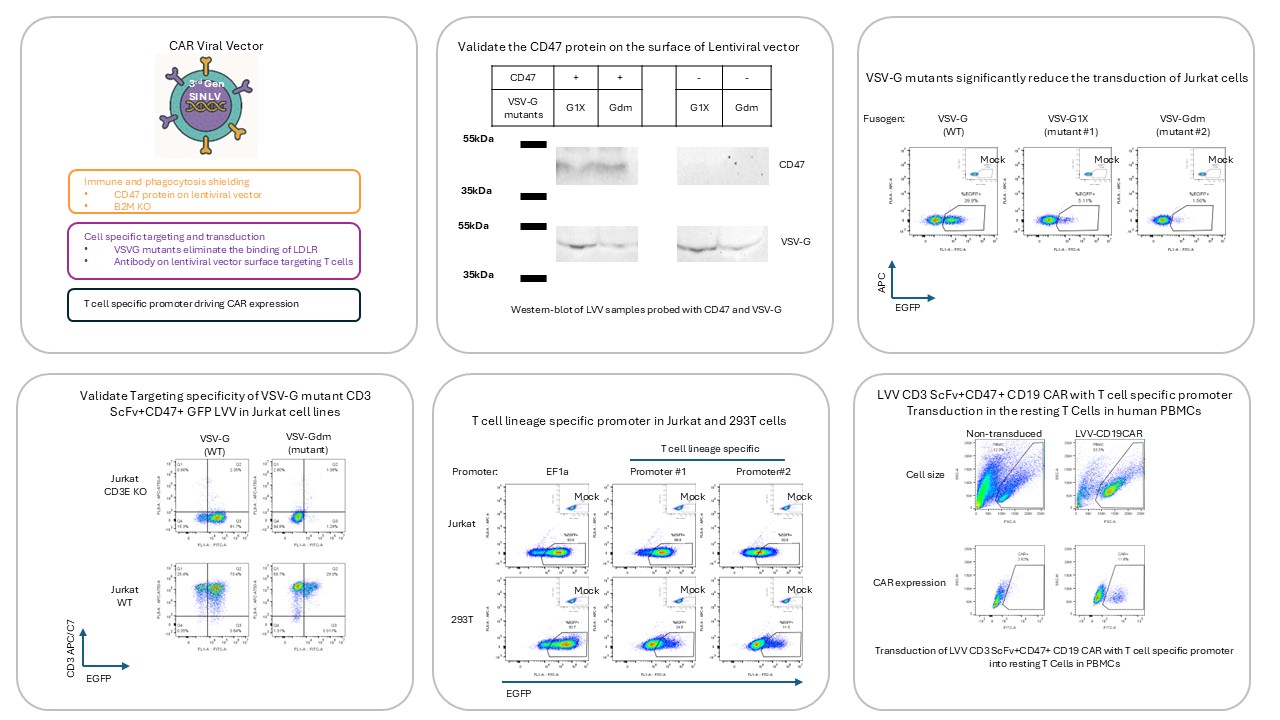
Case Study
Why Choose RedyBio?
- Industry-leading expertise in both non-viral (LNP-RNA) and viral CAR delivery systems
- Deep experience in CAR design and optimization
- Flexible and scalable platforms tailored to your therapeutic goals
Contact us
- info@redybio.com
- (858) 876-8688
- 6331 Nancy Ridge Dr. Suite A San Diego, CA 92121
Let us work together
Reach out for more information or a quote. Our team is here to assist you with your research needs and inquiries.